UDC: 616.37 – 089.87
Vyacheslav Kalentjev1, Dmitry Chichevatov2, Artem Glukhov2
1Penza Regional Oncology Clinical Dispensary, Department of Surgery, 37a prospekt Stroiteley, Penza, 440066, Russia
2Penza State University, Department of Surgery, 40 Krasnaya Street, Penza, 440026, Russia
Dmitry Chichevatov – MD, Professor, Department of Surgery 40 Krasnaya St., Penza 440026, Russian Federation, tel.: +7-927-366-35-85, e-mail: chichevatov69@mail.ru, ORCID ID: 0000-0001-6436-3386
Abstract. Outcomes of pancreaticoduodenectomies (PDE) and total pancreaticoduodenectomies (TPDE) may depend on specific surgery-related anatomical, technical, and postoperative factors. The research evaluates the impact of some of these factors on short- and long-term outcomes.
Material and methods. We analyzed treatment outcomes of 182 patients who underwent PDE/TPDE (92.9% of them for periampullary tumors).
Results. Concomitant multi-visceral resections were not associated with a higher postoperative morbidity (р = 0.516) and mortality (р = 0.153) rates. Angioplastic surgery also did not increase postoperative morbidity (р = 1.000) and mortality (р = 0.325) but led to lower overall survival in patients with pancreatic (HR = 3.03) and duodenal (HR = 4.43) carcinomas. Postoperative complications significantly affected overall survival in patients with a duodenal cancer (HR = 3.24) (p < 0.05). PDEs performed for “non-pancreatic” periampullary carcinomas were associated with soft texture of a pancreas (р = 0.000), a higher rate of complications of class III and above according to Clavien–Dindo classification (р = 0.013), and postoperative pancreatic fistulas (POPF) of types B and C (р = 0.007). The lowest rate of POPF was registered after “duct to mucosa” pancreatic anastomoses (6.6%) compared to invaginational (16.0%) and catheter-guided (20.0%) ones.
Conclusion. Rational planning of treatment for periampullary tumors involves considering specific factors that can influence the overall outcome.
Key words: pancreaticoduodenectomy, total pancreaticoduodenectomy, postoperative pancreatic fistula, porto-mesenteric venous resection, periampullary tumors.
Introduction
Pancreaticoduodenectomy (PDE) or total pancreaticoduodenectomy (TPDE) are principal curative options for periampullary tumors and, above all, a pancreatic carcinoma. Despite some advances in drug therapy, the best long-term results come from a combination of surgery and drug therapy [1]. Nonetheless, PDE/TPDE, as one of the most complex surgeries, may be succeeded by series of severe, even life-threatening specific complications, among which the most important are bleeding and postoperative pancreatic fistula (POPF) [2, 3].
Commonly, such complications prevent adjuvant chemotherapy (ACT), which, in turn, significantly decreases overall survival. The study of Merkow R et al., (2014) [4] showed that in the group of patients with serious postoperative complications ACT was performed more rarely than in the group of uncomplicated operations (43.6% versus 61.8%), with the risk of refusing from ACT increasing more than twofold (OR = 2.20). This issue is so crucial that neoadjuvant chemotherapy (NACT) is currently being discussed not only from an oncological perspective, but also as a way to perform drug treatment before the development of possible surgical complications, that is, to accomplish the treatment protocol [5]. Ultimately, a complex question arises regarding the rational sequence of medical and surgical stages in order to achieve the highest overall survival (OS) taking into account the specific features of surgery. The aim of this study was to evaluate the impact of technical details of surgeries and postoperative complications on short- and long-term outcomes.
Material and methods
We analyzed treatment outcomes of 182 patients who underwent PDE/TPDE for periampullary tumors and non-tumor diseases of a pancreas. Inclusion criteria: patients who underwent PDE. Exclusion criteria: PDE performed as a multi-visceral resection for primary invasive non-periampulary tumor. Patient characteristics are presented in Table 1.
Table 1. Patient characteristics
| Characteristic (data format) | Value |
| Age (min – max; mean ± SD), years | 36 – 80; 62.6 ± 9.6 |
| Sex (males/females) | 103 (56.6%)/79 (43.3%) |
| Diagnosis (n, %) | |
| DCBDC | 8 (4.4%) |
| PNET | 5 (2.7%) |
| AC | 10 (5.5%) |
| DC | 36 (19.8%) |
| PDAC | 104 (57.1%) |
| Other malignancies | 2 (1.1%) |
| Benign tumors | 4 (2.2%) |
| Pancreatitis | 13 (7.1%) |
| Т (is – 1 – 2 – 3 – 4) | 1 – 12 – 21 – 99 – 31 |
| N (0 – 1 – 2) | 105 – 55 – 4 |
DCBDC – distal common bile duct cancer, PNET – pancreatic neuroendocrine tumor,
PDAC – pancreatic ductal adenocarcinoma, AC – ampullary cancer, DC – duodenal cancer
The major part of diagnoses was PDAC and DC. Other malignancies included duodenal lymphoma and the apical lymph node metastasis from colorectal cancer, invading the duodenum and pancreas. Inflammatory diseases and benign tumors accounted for up to 10% of the pathologies. Among malignant tumors, 14 (7.7%) were of stage IV, 13 of them by M1 criterion (solid carcinomas). The treatment characteristics are presented in Table 2.
Table 2. Surgical details, chemotherapy
| Characteristic (data format) | Value |
| Surgery (n, %) | |
| TPDE | 16 (8.8%) |
| PDE | 166 (91.2%) |
| Multi-visceral resections (n, %) | 10 (5.5%) |
| Colon | 5 (2.7%) |
| Liver | 2 (1.1%) |
| Gastrectomy | 2 (1.1%) |
| Small Intestine | 1 (0.5%) |
| Vascular resections (n, %) | 47 (25.8%) |
| Porto-mesenteric venous resection | 41 (22.5%) |
| Resection of arteries | 10 (5.5%) |
| Pancreatic parenchyma density (n, %) | |
| Firm | 123 (67.6%) |
| Soft | 42 (23.1%) |
| Not analyzed | 17 (9.3%) |
| Type of pancreatic anastomosis (n, %) | 165 (90.7%) |
| “Duct to mucosa” | 122 (67.0%) |
| Invaginational | 25 (13.7%) |
| Catheter-guided | 9 (4.9%) |
| Sleeve-shaped (original) | 9 (4.9%) |
| Chemotherapy (n (%), min – max courses)* | 83 (50.9%) |
| NACT | 7 (4.3%), 4 – 8 |
| ACT | 73 (44.8%), 1 – 11 |
| PCT | 3 (1.8%), 8 – 12 |
PCT – perioperative chemotherapy
* — information about 163 patients with PDAC, DC, DCBDC, AC, PNET
PDE was a predominant type of surgery. Multi-visceral resections were combined with vascular resections in 7 cases, i.e. there were a total of 50 (27.5%) combined resections of vessels and other organs.
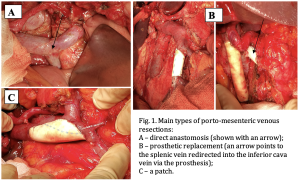
Five types of porto-mesenteric venous resections were performed: 1) without restoring (ligation) (1); 2) marginal resection (9); 3) with a patch application (2); 4) with a direct anastomosis (13); 5) with prosthetic replacement (16). The main types of venous resections are shown in Figure 1.
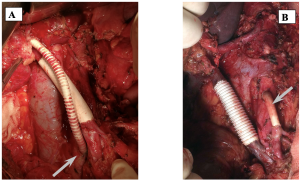
Resections of the hepatic artery (6) and superior mesenteric artery (6) were performed in 10 patients. There were 5 prosthetic replacements, 4 direct anastomoses, and one patch. Variants of arterial angioplasties are shown in Figure 2.

The density of the pancreatic parenchyma was assessed clinically: visually and by palpation, without the use of additional densitometry methods. The main criterion of the “soft” parenchyma was easy cutting of a surgical suture through. In 17 cases, the pancreatic parenchyma density was not assessed because these were 16 TPDE and 1 PDE without an anastomosis when the density did not matter.
During the PDE procedures, 4 types of pancreatic anastomoses were performed: 1) “duct to mucosa” technique with precise suturing of the pancreatic duct into the intestinal loop (n = 122); 2) invaginational anastomoses when the entire proximal part of the pancreatic stump was sutured into the jejunum (n = 25); 3) “catheter-guided” anastomoses with catheterization of the pancreatic duct and forming an external preventive pancreaticostomy (n = 9); 4) a “sleeve-shaped” anastomosis, which involved passing the pancreatic stump through a window in the mesentery of the small intestine, forming a tight small intestinal sleeve around the pancreatic stump with precise suturing of the pancreatic duct (invention application No. 2025110761/14(026610)) (n = 9). The standard types of pancreatic anastomoses were classified according to Olakowski M et al., (2020) [6].
Information on chemotherapy is presented only for PDAC, DC, AC, PNET, and DCBDC (n = 163). Eight different regimens of chemotherapy were employed. The most commonly used were FOLFIRINOX, gemcitabine-based regimens (GEM, GEMCAP, GEMOX), LF, and capecitabine.
Statistical methods
All statistical calculations were made using the freely distributable ‘R’ programming language (v. 4.4.1, Free Software Foundation), ‘RStudio’ integrated developer media (2024.09.1 Build 394 © 2009-2024 Posit Software, PBC), and ‘JASP’ statistical package (v. 0.18.3, Built on Jan 11 2024, © 2013-2024 University of Amsterdam). The probability of the type I error of p = 0.05 was regarded as the threshold of statistical significance. Descriptive statistics included a mean, median, standard deviation (SD), and a standard error (SE); a 95% confidence interval (95% CI) was used. Fisher’s exact test and Pearson’s χ2 test were applied to analyze contingency tables. We performed Bayesian regression analysis using the ‘brms’ R library (Bayesian Regression Models using ‘Stan’, v. 2.21.0, https://github.com/paul-buerkner/brms). Survival analysis was performed using the ‘survival’ (v. 3.6-4, https://github.com/therneau/survival) and ‘survminer’ (Drawing Survival Curves using ‘ggplot2’, v. 0.4.9, https://rpkgs.datanovia.com/survminer/index.html) R libraries. Survival curves were compared using the log-rank test. A graphical analysis was performed using the ‘ggplot2’ R library (Create Elegant Data Visualisations Using the Grammar of Graphics, v. 3.5.1, https://ggplot2.tidyverse.org).
Results
Immediate outcomes of surgical procedures
A total of 88 postoperative complications appeared in 83 patients (45.6%), i.e. five subjects experienced 2 complications each. The structure of postoperative complications is shown in Table 3.
Table 3
Structure of postsurgical complications
| Complication | Rate | Percentage |
| DGE | 24 | 13.2% |
| POPF C | 11 | 6.0% |
| Pancreatitis\pancreonecrosis | 8 | 4.4% |
| Pneumonia | 6 | 3.3% |
| Internal bleeding | 4 | 2.2% |
| External bleeding | 6 | 3.3% |
| Eventration | 4 | 2.2% |
| POPF B | 4 | 2.2% |
| Bile leakage | 5 | 2.7% |
| Renal failure | 2 | 1.1% |
| Cholangitis | 2 | 1.1% |
| Abdominal abscess | 1 | 0.5% |
| Myocardial infarction | 1 | 0.5% |
| Mesenteric artery thrombosis | 1 | 0.5% |
| Wound infection | 1 | 0.5% |
| Left liver lobe necrosis | 1 | 0.5% |
| Liver failure | 1 | 0.5% |
| Acute gastric ulcer | 1 | 0.5% |
| Colon anastomosis leakage | 1 | 0.5% |
| Afferent loop syndrome | 1 | 0.5% |
| Prosthesis thrombosis | 1 | 0.5% |
| Diarrhea | 1 | 0.5% |
| Retroperitoneal phlegmon | 1 | 0.5% |
| Total | 88 |
DGE – delayed gastric emptying
The table shows that complications specific to PDE predominated, with DGE being the most common. There were only 15 clinically relevant POPF of types B and C (8.2%). All internal bleedings were not associated with POPF; the origins of external bleedings were acute gastric ulcers and, in one case, the pancreatic anastomosis area. Pancreatites and pancreonecroses of the stump had relatively high rate. Complications specific to angioplasty occurred only once (prosthesis thrombosis). All other complications were characteristic to abdominal surgery; many of them were single.
According to the Clavien-Dindo classification, postoperative complications were as follows: class II – 41 (22.5%), III – 22 (12.1%), IV – 7 (3.8%), V – 13 (7.1%). Four patients died of POPF of type C, 2 – of pancreatic necrosis. External bleeding, peritonitis due to intestinal anastomosis leakage, retroperitoneal phlegmon, mesenteric artery thrombosis, myocardial infarction, and liver necrosis were causes of death of one patient for each case. Thus, POPF accounted for the largest number of fatal outcomes. In the POPF group, the mortality rate was four out of 15 (26.7%).
Impact of technical features of surgery on the immediate outcomes
In this study, we assumed that certain technical details of surgeries, increasing their trauma, duration, and distinguishing them from the standard extents, could directly or indirectly influence the treatment outcome. Multi-visceral and vascular resections, pancreatic parenchyma texture, and a type of pancreatic anastomosis were considered such factors. The latter two were analyzed because they are directly related to POPF, the most frequent, specific, potentially fatal or disabling complication that prevents from accomplishing the full treatment protocol.
In the group of multi-visceral resections, complications were observed in 6 patients out of 10; in the group where resection of other organs was not performed, 77 complications were reported in 172 patients (44.8%). This difference was not statistically significant (p = 0.516). Postoperative mortality was 2 out of 10 versus 11 out of 172 (6.4%), respectively. Although mortality rate was higher in the group of multi-visceral resections, the difference was statistically insignificant (p = 0.153) either.
In the angioplasty group, post-operative complications were observed in 21 cases out of 47 (44.7%); among 135 patients without angioplasty, 62 complications (45.9%) were recorded, with no significant difference (p = 1.000). Postoperative mortality in the angioplasty group was 5 of 47 (10.6%) versus 8 of 135 (5.9%) in the standard group. The difference in the rate was also statistically insignificant (p = 0.325).
In order to evaluate the influence of a pancreatic anastomosis type as a technical specificity of the operation, which can cause a potentially fatal complication of clinically relevant POPF, a regression model was made (Table 4).
Table 4. Regression coefficients. Influence of the type of pancreatic anastomosis on POPF incidence
| Family: bernoulli; Links: mu=logit; Formula: fistula ~ 1 + (1|anastomosis);
Data: df (Number of observations: 165) |
|||||||
| Draws: 4 chains, each of iterations=2000; warmup=1000; thin=1; total post-warmup draws=4000 | |||||||
| Multilevel Hyperparameters: ~ anastomosis (Number of levels: 4) | |||||||
| Estimate | SE | l-95% | u-95% | Ȓ | Bulk_ESS | Tail_ESS | |
| sd (Intercept) | 1.19 | 1.06 | 0.08 | 4.20 | 1.00 | 810 | 1088 |
| Individual intercepts: | |||||||
| Intercept (type 1 “duct-to-mucosa”) | –2.59 | 0.34 | –3.28 | –1.94 | |||
| Intercept (type 2 “invaginational”) | –1.94 | 0.52 | –2.99 | –0.92 | |||
| Intercept (type 3 “catheter guided”) | –1.81 | 0.72 | –3.17 | –0.37 | |||
| Intercept (type 4 “sleeve-shaped”) | –3.04 | 1.37 | –6.68 | –1.31 | |||
| Regression coefficients: | |||||||
| Intercept | –2.15 | 0.79 | –3.78 | –0.38 | 1.00 | 762 | 634 |
Ȓ — potential scale reduction factor; Bulk_ESS – effective sample size for the central part of the posterior distribution; Tail_ESS – effective sample size for the tail parts of the posterior distribution;
The actual multilevel regression model did not contain any predictors, but only calculated the expected average rate of POPF (Intercept) and rates by groups corresponding to the types of anastomoses (Individual Intercepts). Based on this model, the expected frequencies of POPF were calculated depending on the type of anastomosis: 1) “duct-to-mucosa” – 6.9% [3.6–12.6]; 2) invaginational anastomosis – 12.6% [4.8–28.5]; 3) “catheter guided” – 13.9% [3.9–40.9]; 4) “sleeve-shaped” – 4.6% [0.1–21.2]. Taking into account the confidence intervals, the calculated estimates coincided with those observed in the sample (6.6%, 16.0%, 22.0%, 0.0% respectively).
Another well-known technical feature that could affect the POPF rate was the pancreatic parenchyma density. In the present study, 14 out of 42 (33.3%) patients with a “soft” pancreas developed POPF and none of 123 patients with a firm parenchyma pancreas (p = 0.000) did. One patient with type B fistula was not included in the analysis because the PDE was performed without a pancreatic anastomosis (a clogged stump of the pancreas including only the tail of the organ). It should be noted that the pancreas texture was also associated with the number of complications in general: 49 out of 123 (39.8%) complications were observed in the group of firm glands, 26 out of 42 (61.9%) –in the group of soft glands (p = 0.019). Factors of the surgery technological specificity and their influence on the immediate outcome according to the tumor type are shown in Table 5.
Table 5. Factors of PDE surgery specificity
| Diagnosis | n* | «Soft» PG | Clavien–Dindo
of class III and > |
POPF
of types В and С |
Died |
| PDAC | 89 | 9 (10.1%) | 14 (15.7%) | 4 (4.5%) | 4 (4.5%) |
| DC | 36 | 13 (36.1%) | 9 (25.0%) | 3 (8.3%) | 5 (13.9%) |
| AC | 10 | 8 (80.0%) | 5 (50.0%) | 2 (20.0%) | 0 |
| DCBDC | 8 | 7 (87.5%) | 5 (62.5%) | 3 (37.5%) | 0 |
| PNET | 3 | 1 (33.3%) | 0 | 0 | 0 |
| Total | 146 | 38 (26.0%) | 33 (22.6%) | 12 (8.2%) | 9 (6.2%) |
| Comparison of all types of tumors | |||||
| р | 0.000 | 0.013 | 0.007 | 0.533 | |
| Comparison of tumors excluding PDAC | |||||
| p | 0.000 | 0.080 | 0.102 | 0.664 | |
* – PDE for primary periampullary malignant tumors only are presented
Thus, Table 5 shows that statistically significant factors in the PDE group for PDAC were as follows: the minimal number of “soft” pancreatic glands, the lowest number of major complications by Clavien–Dindo classification, and the lowest number of POPF of types B and C. PDE for all other periampullary tumors differed significantly only in terms of further increase in the number of “soft” pancreatic glands.
Influence of technical features of surgeries and post-operative complications on the long-term outcome of treatment
In this section, only PDAC and DC were analyzed because all other tumors were found in limited or isolated observations. Initially, the Cox regression model (Table 6) was developed by sequential entering of predictors for overall survival in PDAC.
Table 6. Cox regression model for PDAC
| Predictor | β | HR | SE | Z | p |
| Chemotherapy (yes) | –1.17 | 0.31 | 0.46 | –2.56 | 0.010 |
| Angioplasty (no) | –1.12 | 0.33 | 0.32 | –3.48 | 0.000 |
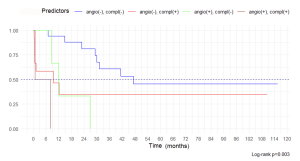
Only chemotherapy and angioplasty affected the overall survival in the PDAC group. Multi-visceral resections (yes, no) and postoperative complications (yes, no) had regression coefficients not differing from zero (р > 0.100) in this model. Chemotherapy was a significant predictor only if 4 courses of NACT, or 3 courses of ACT, or monotherapy with capecitabin were regarded. Kaplan–Meier curves are shown in Figure 3.
|
The best results were obtained in the group of patients without angioplasty but with chemotherapy, the median of OS was 29.9 mo [20.9 – 51.1], 5‑year OS – 29.8% [16.4–54.1]. The results were relatively worse in the group where chemotherapy was not used: the median of OS was 22.6 mo [13.6 – 53.3], 5‑year OS – 9.3% [2.5–34.8]. In the group of angioplasty combined with chemotherapy, the median of OS was 13.5 mo [9.5–26.1], all patients died within 50 mo. In the group of angioplastic surgery, only the median of OS was 4.9 mo [2.7 – NA], all patients died within 12 mo.
In patients with postoperative complications, ACT was administered in 32 of 50 (64.0%) versus 36 of 54 (66.7%) in the non-complicated group (р = 0.838). Chemotherapy was applied in 15 of 18 (83.3%) patients who had DGE versus 53 of 86 (61.7%) who did not have this specific complication (р = 0.104). Hence, postoperative complications did not influence ACT significantly in the PDAC group.
The same analysis was performed for patients with DC. We could not assess the effect of multi-visceral resections as in the DC group there was only one such a procedure. Unlike PDAC, chemotherapy had no impact on OS in DC cancer; the regression coefficient for this predictor was not significantly different from 0 (p = 0.955). Thus, only angioplasty and the postoperative complications had a significant effect on OS (Table 7).
Table 7. Cox regression model for DC
| Predictor | β | HR | SE | Z | p |
| Postoperative complications (yes) | 1.17 | 3.24 | 0.47 | 2.50 | 0.013 |
| Angioplasty (yes) | 1.49 | 4.43 | 0.57 | 2.60 | 0.009 |
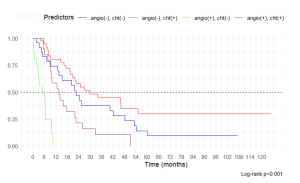
Kaplan–Meier curves are shown in Figure 4.
Fig. 4. Kaplan–Meier curves for 4 groups of patients with DC depending on angioplasty and postoperative complications
Pairwise comparison of curves revealed that only the group where there was no angioplasty and post-operative complications significantly had the best OS compared to all other groups (p < 0.05); the rest of the groups were not pairwise different (p > 0.05). In the group of surgery without angioplasty and without complications, the median of OS was 47.3 mo [30.0–NA], 5-year OS – 45.6% [25.8–80.6]. The results were worse in the group without angioplasty, but with postoperative complications: the median of OS was 9.5 mo [0.7–NA], 5-year OS – 35.0% [14.8–82.8]. In the group with angioplasty and without complications, the median of OS was 11.9 mo [8.5–NA], all patients died within 26.8 months. In the group of complicated angioplastic surgery, the median of OS was 4.4 mo [0.8–NA], all patients died within 8 months.
Discussion
In this study, we considered a number of factors that may affect the overall treatment of periampullary tumors. All these factors, combined by the generalizing term “specificity” of PDE and TPDE, represented anatomo-physiological features of the patient, technical details of the surgery, and post-operative complications. We assumed that «specificity» could determine the outcome and a long-term forecast, therefore its analysis may help to reveal resources to improve the result. The study did not analyze commonly known or well-studied factors such as sex, age, TNM, stage and morphology of the tumor because they are nonspecific; we emphasized specific technological aspects of PDE and TPDE.
Since PDE/TPDEs have not been standardized, easily replicable interventions yet, their technical complexity (multi-visceral resections) may provoke a higher risk to the patient. In the present study, this assumption was not confirmed either in terms of complications rate or in terms of post-operative mortality. Other authors consider the PDE/TPDE with multi-visceral resection to be a well-justified procedure requiring a very balanced approach, as the complications rate can reach 56–69%, and mortality rate – 7–10% [7, 8].
Apparently, resection and plastic surgery of vessels should be considered as a PDE/TPDE-specific extended option. This is due to the fact that, first, it is the basis for assessing the tumor resectability [9]; second, vascular tumor invasion determines the tumor stage and prognosis. According to the results of this study, resection and angioplasty did not significantly affect the short-term outcomes of surgery: the number of complications and mortality rate were comparable with a group of standard surgery. Overall survival, by contrast, distinguished significantly (Figures 3 and 4). Angioplasty was a marker of a poor prognosis in both DC and PDAC, although in the latter group 4-year OS was achieved in NACT/ACT. Thus, vascular resection should be considered more in the oncological rather than technological aspect. In the recent decade, the feasibility of angioplastic PDE/TPDE has been widely discussed. Some authors believe that this is a way to improve overall survival and long-term results [10]. Other authors mention the specific complications of angioplasty, like acute porto-mesenteric thrombosis, which can increase mortality from 4.6% to 22.2% and decrease OS median from 15.9 down to 7.1 months in PDAC [11]. In the present study, only one early thrombosis of the prosthesis was not followed by adverse events. We did not investigate late thromboses, although they may be of prognostic significance [12]. With 4-year survival in the group of angioplasty surgery combined with CT, we consider vascular resection in PDAC to be reasonable.
Postoperative complications had different prognostic significance depending on the particular tumor type: they were not a predictor of poor prognosis in PDAC and, on the contrary, had statistically significant effect in PDE for DC. Analysis of survival curves (Fig. 3, 4) shows rapid decline in groups of complicated PDE for DC. One should note the small value of the OS median (9.5 months) and steady 5-year OS at 35.0% in the group of complicated PDE without angioplasty for DC. Thus, the negative impact of complications in DC consisted in a large number of deaths in the earliest period of observation, which was not the case with PDAC or uncomplicated PDE for DC.
Table 5 largely explains this phenomenon. Postoperative complications of class III and above (Clavien–Dindo) were concentrated in the group of PDE, performed not for PDAC. In this group, 8 out of 15 POPF, 5 out of 8 pancreatites/pancreonecroses, 3 out of 5 bile leakages, 3 out of 6 external bleedings were registered. Since there were no statistically significant differences within this group (Table 5), conditionally all PDE could be divided into “pancreatic” (for PDAC) and “non-pancreatic” (for other periampullary tumors) ones.
Table 5 also shows that the group of “non-pancreatic” PDE was statistically significantly a group with predominantly “soft” pancreatic glands and higher rate of POPF. Traditionally, contemporary publications widely discuss “soft” parenchyma of PG and the absence of pancreatic hypertension influence on POPF development [13-15]. However, the recent large study by Czarnecka Z. et al., (2025) has demonstrated a significant negative influence of PG soft texture on 19 postoperative parameters and characteristics. Among them, there was a higher rate of POPF (threefold), acute kidney injury (twofold), infections of deep spaces and sepsis, wound infection, eventration, pneumonia, and pulmonary embolism [16]. It can be assumed that the true importance of this PDE-specific anatomical characteristic of the patient is currently underestimated.
Clinically relevant POPF of types B and C, however, remain the main specific postoperative complication negatively affecting the outcome. First, it is caused by their high rate, which can vary from 2 to 37% [17, 18]. Second, mortality in this group is as high as 40% [3]. In the present study, among all postoperative complications, POPF ranked 2nd (8.2%) after DGE and they had a mortality rate of 26.7%. Thus, POPF are a major resource for improving the treatment outcomes of periampullary tumors. A reliable pancreatic anastomosis remains the main method among many ways to prevent POPF. Today, many modifications of pancreatic anastomoses are developed and classified [6]; however, the recent large meta-analysis has not demonstrated considerable benefits for their specific types [19]. The POPF rate remains high; Vining C et al. (2020) reported 15.6% of fistulas after 12,612 open PDE [20]. In the present study, we applied 3 worldwide well-known types of pancreatic anastomosis and 1 original (“sleeve-shaped”) type. Regression analysis (Table 4) showed poor results of using invaginational and catheter-guided anastomoses, which led to their disuse. The acceptable POPF rate was after “duct to mucosa” anastomosis, but only in the case of the firm pancreatic gland (0 POPF out of 114 operations); this anastomosis was completely inefficient in a soft gland (8 POFF out of 8 operations). The original anastomosis was applied only in “soft” parenchyma of PG; POPF were not registered. Given the small number of observations (n = 9), it is premature to assess the efficacy of this anastomosis; however, regression modeling shows the lowest expected rate of POPF with this anastomosis (4.6%).
Limitations
Insufficient number of observations was a factor limiting the study. An informative survival analysis was possible only in DC and PDAC. The analysis of long-term outcomes in other periampullary tumors was inconclusive; however, they were not excluded from the study because they made an informative contribution to the evaluation of short-term outcomes.
The inability of randomization and control in assessing the efficiency of pancreatic anastomoses also limited the study, and did not allow carrying out direct comparative analysis. The invaginational and catheter-guided anastomoses immediately demonstrated a high rate of POPF, so the patients were not included in these groups for ethical reasons.
Conclusion
Currently, the short- and long-term outcomes of PDE/TPDE are complex in nature and may depend on a number of anatomical, clinical, technical and postoperative factors specific to these operations. Angioplasty, which has entered the practice of oncological surgical pancreatology, is accompanied by more modest long-term results compared to standard surgery and its application requires further study. Specific postoperative complications, such as POPF, as well as class III and above by Clavien–Dindo classification, in general, worsen both the surgery outcome and the long-term prognosis. Performing a surgery taking into account the anatomical specifics of the pancreas, developing and reasonably choosing a pancreatic anastomosis can contribute to the prevention of these complications and improve treatment results.
References
- Stocken DD, Büchler MW, Dervenis C, Bassi C, Jeekel H, Klinkenbijl JH, Bakkevold KE, Takada T, Amano H, Neoptolemos JP; Pancreatic Cancer Meta-analysis Group. Meta-analysis of randomised adjuvant therapy trials for pancreatic cancer. Br J Cancer. 2005 Apr 25;92(8):1372-81. doi: 10.1038/sj.bjc.6602513. PMID: 15812554; PMCID: PMC2361989.
- Bassi C, Marchegiani G, Dervenis C, Sarr M, Abu Hilal M, Adham M, Allen P, Andersson R, Asbun HJ, Besselink MG, Conlon K, Del Chiaro M, Falconi M, Fernandez-Cruz L, Fernandez-Del Castillo C, Fingerhut A, Friess H, Gouma DJ, Hackert T, Izbicki J, Lillemoe KD, Neoptolemos JP, Olah A, Schulick R, Shrikhande SV, Takada T, Takaori K, Traverso W, Vollmer CM, Wolfgang CL, Yeo CJ, Salvia R, Buchler M; International Study Group on Pancreatic Surgery (ISGPS). The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 Years After. Surgery. 2017 Mar;161(3):584-591. doi: 10.1016/j.surg.2016.11.014. Epub 2016 Dec 28. Erratum in: Surgery. 2024 Sep;176(3):988-989. doi: 10.1016/j.surg.2024.05.043. PMID: 28040257.
- Загайнов В.Е., Киселев Н.М., Колесник Я.И., Кучин Д.М. Сосудистые осложнения панкреатодуоденальной резекции, их профилактика и лечение. Вопросы онкологии. 2024; 70(2): 360-367.-DOI:10.37469/0507-3758-2024-70-2-360-367. [Vadimir E. Zagainov, Nikolai M. Kiselev, Yan I. Kolesnik, Denis M. Kuchin. Vascular complications of pancreaticoduodenal resection: prevention and treatment. Voprosy Onkologii = Problems in Oncology. 2024; 70(2): 360-367. (In Rus).-DOI: 10.37469/0507-3758-2024-70-2-360-367].
- Merkow RP, Bilimoria KY, Tomlinson JS, Paruch JL, Fleming JB, Talamonti MS, Ko CY, Bentrem DJ. Postoperative complications reduce adjuvant chemotherapy use in resectable pancreatic cancer. Ann Surg. 2014 Aug;260(2):372-7. doi: 10.1097/SLA.0000000000000378. PMID: 24374509.
- Вервекин И.В., Захаренко А.А. Непосредственные результаты применения неоадъювантной химиотерапии по схеме mFOLFIRINOX у больных резектабельным раком поджелудочной железы. Сибирский онкологический журнал. 2025; 24(4): 43–53. – doi: 10.21294/1814-4861-2025-24-4-43-53. [Vervekin I.V., Zakharenko A.A. Immediate results of neoadjuvant chemotherapy with mFOLFIRINOX regimen in patients with resectable pancreatic cancer. Siberian Journal of Oncology. 2025; 24(4): 43–53. – doi: 10.21294/1814-4861-2025-24-4-43-53].
- Olakowski M, Grudzińska E, Mrowiec S. Pancreaticojejunostomy — a review of modern techniques. Langenbecks Arch Surg. 2020 Feb;405(1):13-22. doi: 10.1007/s00423-020-01855-6. Epub 2020 Jan 23. PMID: 31975148; PMCID: PMC7036071.
- Petrucciani N, Debs T, Nigri G, Giannini G, Sborlini E, Kassir R, Ben Amor I, Iannelli A, Valabrega S, D’Angelo F, Gugenheim J, Ramacciato G. Pancreatectomy combined with multivisceral resection for pancreatic malignancies: is it justified? Results of a systematic review. HPB (Oxford). 2018 Jan;20(1):3-10. doi: 10.1016/j.hpb.2017.08.002. Epub 2017 Sep 22. PMID: 28943396.
- Schwartz PB, Roch AM, Han JS, Vaicius AV, Lancaster WP, Kilbane EM, House MG, Zyromski NJ, Schmidt CM, Nakeeb A, Ceppa EP. Indication for en bloc pancreatectomy with colectomy: when is it safe? Surg Endosc. 2018 Jan;32(1):428-435. doi: 10.1007/s00464-017-5700-0. Epub 2017 Jun 29. PMID: 28664444.
- Isaji S, Mizuno S, Windsor JA, Bassi C, Fernández-Del Castillo C, Hackert T, Hayasaki A, Katz MHG, Kim SW, Kishiwada M, Kitagawa H, Michalski CW, Wolfgang CL. International consensus on definition and criteria of borderline resectable pancreatic ductal adenocarcinoma 2017. Pancreatology. 2018 Jan;18(1):2-11. doi: 10.1016/j.pan.2017.11.011. Epub 2017 Nov 22. PMID: 29191513.
- Melo Araújo de Mendonça CM, Krutman M, Nishinari K, Pignataro BS, Chojniak R, Cavalcante RN, Centofanti G, Inforsato N, Yazbek G. Survival and patency outcomes in a 10-year experience of cephalic pancreatectomy associated with portomesenteric venous reconstruction. Ann Vasc Surg. 2025 May 23:S0890-5096(25)00371-1. doi: 10.1016/j.avsg.2025.05.027. Epub ahead of print. PMID: 40414532.
- Gawlas I, Epelboym I, Winner M, DiNorcia J, Woo Y, Lee JL, Schrope BA, Chabot JA, Allendorf JD. Short-term but not long-term loss of patency of venous reconstruction during pancreatic resection is associated with decreased survival. J Gastrointest Surg. 2014 Jan;18(1):75-82. doi: 10.1007/s11605-013-2375-2. Epub 2013 Oct 10. PMID: 24114682.
- Ishida H, Watanabe S, Rodriguez Franco S, Franklin O, Stoop TF, Kirsch MJ, Schulick RD, Del Chiaro M. Portomesenteric Venous Thrombosis after Pancreatic Cancer Surgery with Venous Resection and Reconstruction: Timing, Incidence, and Risk Factors. Ann Surg. 2024 Dec 16. doi: 10.1097/SLA.0000000000006605. Epub ahead of print. PMID: 39676646.
- Коваленко З.А., Ефанов М.Г. Шкалы прогноза панкреатической фистулы после операции Уиппла. Хирургия. Журнал им. Н.И. Пирогова. 2021;07:71–76. https://doi.org/10.17116/hirurgia202107171. [Kovalenko ZA, Efanov MG. Scoring systems to predict pancreatic fistula after Whipple procedure. Pirogov Russian Journal of Surgery = Khirurgiya. Zhurnal im. N.I. Pirogova. 2021;07:71–76. (In Russ.). https://doi.org/10.17116/hirurgia202107171].
- Суворов В.А., Панин С.И., Коваленко Н.В., Жаворонкова В.В., Постолов М.П., Толстопятов С.Е., Бубликов А.Е., Панова А.В., Попова В.О. Прогнозирование панкреатической фистулы после панкреатодуоденальной резекции с использованием машинного обучения. Сибирский онкологический журнал. 2023; 22(6):25–34. – doi: 10.21294/1814-4861-2023-22-6-25-34. [Suvorov V.A., Panin S.I., Kovalenko N.V., Zhavoronkova V.V., Postolov M.P., Tolstopyatov S.E., Bublikov A.E., Panova A.V., Popova V.O. Prediction of pancreatic fistula after pancreatoduodenectomy using machine learning. Siberian Journal of Oncology. 2023; 22(6): 25–34. – doi: 10.21294/1814-4861-2023-22-6-25-34].
- Chen G, Zheng Z, Yi H, Yue Q, Li L. An analysis of risk factors for clinically relevant pancreatic fistulas after laparoscopic pancreaticoduodenectomy. Medicine (Baltimore). 2023 May 19;102(20):e33759. doi: 10.1097/MD.0000000000033759. PMID: 37335734; PMCID: PMC10194647.
- Czarnecka Z, Verhoeff K, Bigam D, Dajani K, Shapiro J, Anderson B. Impact of soft pancreas on pancreaticoduodenectomy outcomes and the development of the preoperative soft pancreas risk score. Ann Hepatobiliary Pancreat Surg. 2025 Feb 28;29(1):62-71. doi: 10.14701/ahbps.24-172. Epub 2024 Dec 2. PMID: 39617392; PMCID: PMC11830892.
- Neupane HC, Tamang TY, Timalsina S, Tamrakar KK, Bhattarai A. Complications among Patients Undergoing Pancreaticoduodenectomy in Tertiary Care Centers of Nepal: A Descriptive Cross-sectional Study. JNMA J Nepal Med Assoc. 2022 Jan 15;60(245):77-82. doi: 10.31729/jnma.7050. PMID: 35199675; PMCID: PMC9157660.
- Quero G, Menghi R, Fiorillo C, Laterza V, De Sio D, Schena CA, Di Cesare L, Cina C, Longo F, Rosa F, Alfieri S. The impact of gastrojejunostomy orientation on delayed gastric emptying after pancreaticoduodenectomy: a single center comparative analysis. HPB (Oxford). 2022 May;24(5):654-663. doi: 10.1016/j.hpb.2021.09.015. Epub 2021 Sep 24. PMID: 34654621.
- Hai H, Li Z, Zhang Z, Cheng Y, Liu Z, Gong J, Deng Y. Duct-to-mucosa versus other types of pancreaticojejunostomy for the prevention of postoperative pancreatic fistula following pancreaticoduodenectomy. Cochrane Database Syst Rev. 2022 Mar 15;3(3):CD013462. doi: 10.1002/14651858.CD013462.pub2. PMID: 35289922; PMCID: PMC8923262.
- Vining CC, Kuchta K, Schuitevoerder D, Paterakos P, Berger Y, Roggin KK, Talamonti MS, Hogg ME. Risk factors for complications in patients undergoing pancreaticoduodenectomy: A NSQIP analysis with propensity score matching. J Surg Oncol. 2020 Aug;122(2):183-194. doi: 10.1002/jso.25942. Epub 2020 May 23. PMID: 32445612.
List of abbreviations
PG – pancreatic gland
PDE – pancreaticoduodenectomy
TPDE – total pancreaticoduodenectomy
CT or cht – chemotherapy
ACT, NACT – adjuvant, neoadjuvant chemotherapy
PCT – perioperative chemotherapy
DCBDC – distal common bile duct carcinoma
PNET – pancreatic neuroendocrine tumor
AC – ampullary carcinoma
DC – duodenal carcinoma
PDAC – pancreatic ductal adenocarcinoma
POPF – postoperative pancreatic fistula
DGE – delayed gastric emptying
OR, HR – odds ratio, hazard ratio
OS – overall survival
SE – standard error
SD – standard deviation
CI – confidence interval
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of competing interest
The authors have no conflicts of interest to declare.
Authors’ Information
Vyacheslav Kalentjev, MSc, surgeon, Department of Surgery, Penza Regional Oncology Hospital, 37a prospekt Stroiteley, Penza, 440066, Russia
e-mail: kalentev1980@gmail.com,
Phone: 8-927-385-84-40.
SPIN- 3235-6383.
Dmitry Chichevatov, Dr. Sci. (Medicine), Professor, Department of Surgery, Penza State University, 40 Krasnaya Street, Penza, 440026, Russia
e-mail: chichevatov69@mail.ru,
Phone: 8-927-366-35-85.
Researcher ID: Q-3647-2017,
ORCID: 0000-0001-6436-3386
Artem Glukhov, MSc, Assistant Lecturer, Department of Surgery, Penza State University, 40 Krasnaya Street, Penza, 440026, Russia
e-mail: rossomaxa2008@yandex.ru,
Phone: 8-937-414-16- 21.
SPIN- 9136-7827.
Authorship
Dmitry Chichevatov: the conception and design of the study, analysis and interpretation of data.
Vyacheslav Kalentjev, Artem Glukhov: acquisition of data, analysis and interpretation of data, drafting the article and revising it critically for important intellectual content.
Dmitry Chichevatov, Vyacheslav Kalentjev, Artem Glukhov: final approval of the version to be submitted.